`NCESRTA Capacity Building in Clinical Trials

There is a rising incidence of non-communicable diseases (NCD) globally with sub–Saharan African countries additionally affected by sickle cell disease (SCD), a genetic blood disorder of high prevalence. SCD is a leading cause of mortality in children <5 years as well as in youth 5–14 years and adults 15–49 years. Individuals with SCD are also at increased risk of death from stroke, cardiovascular, renal and pregnancy complications and under-five mortalities.
The Sickle Pan-African Research Consortium (SPARCO) was funded by NHLBI in 2017 to build the infrastructure for SCD management and research in sub-Saharan Africa. In Nigeria, under Sickle Pan-Africa Research Consortium Nigeria Network (SPARC-NEt), 25 clinical sites are involved in the registry of 10,336 patients that have been established and are being prospectively followed up.
Skills development is key to any sustainable capacity building effort, but such undertakings require adaptable and tailored strategies, predicated on identified gaps. In keeping with the World Health Organization (WHO) global action plan for the prevention and control of non-communicable diseases (NCDs) which include sickle cell disease (SCD). there is a need for the promotion and support of national capacity for high-quality research and health system development including clinical trials.
Clinical trials of new therapies for SCD are beginning to trickle into the country, bolstered by the recognized status of Nigeria in terms of the burden of sickle cell disease therefore, it is imperative to deepen the knowledge and expertise of researchers in-country to participate in clinical trials towards the delivery of effective patient-centered care. The purpose of this training grant is to increase the capacity of the health care workers and researchers in the SPARCO Sites to participate in clinical trials to improve SCD patient care and management.
In response to these, IQVIA is entering into partnership with the National Centre of Excellence in Sickle Cell Research and Training (NCESRTA) to provide training in clinical trials aimed at building the capacity of SPARC-NEt trial naïve sites to conduct clinical trials. This training is being carried out in collaboration with Bio Ventures for Global Health (BVGH). Resource persons are drawn from across the world, as well as key regulatory agencies such as National Agency for Food and Drug Control (NAFDAC) and National Health Research Ethics Committee (NHREC) of Nigeria.
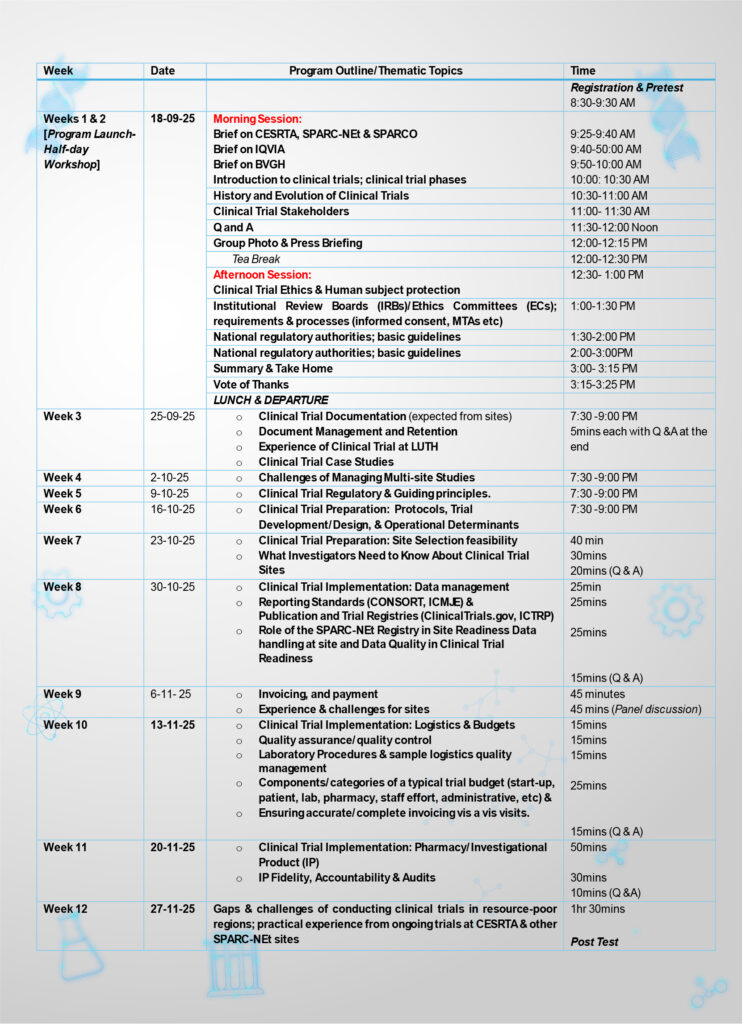
Course Duration: 12 weeks
Delivery Mode/Format: Virtual
Sessions: 1.5 hours weekly
Dates: September 18th – November 27th, 2025
Course Objectives:
By the end of this course, participants will be able to:
- Understand the fundamental concepts of clinical research and trials
- Differentiate between the various phases of clinical trials
- Appreciate the ethical, regulatory, and scientific framework of clinical trials
- Describe the role of stakeholders in the clinical research process
- Understand the design, conduct, and reporting of clinical trials
Past Sessions Recordings
Week 1&2: 12-Week Intensive Clinical Trial Training Course
Week 3: 12-Week Intensive Clinical Trial Training Course
Week 4: 12-Week Intensive Clinical Trial Training Course
Week 5: 12-Week Intensive Clinical Trial Training Course
Week 7: 12-Week Intensive Clinical Trial Training Course
Week 8: 12-Week Intensive Clinical Trial Training Course
Week 9: 12-Week Intensive Clinical Trial Training Course
Week 10: 12-Week Intensive Clinical Trial Training Course
Week 11: 12-Week Intensive Clinical Trial Training Course
Week 12: 12-Week Intensive Clinical Trial Training Course
About us
Twitter Feeds
Copyright © 2024 CESRTA. All rights reserved.

